UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d)
of the Securities Exchange Act of 1934
Date of Report (Date of earliest event reported): February 19, 2026

(Exact name of registrant as specified in its charter)
| (State or other jurisdiction of incorporation) | (Commission File Number) | (IRS Employer Identification Number) | ||||||
(Address of principal executive offices, including Zip Code)
Registrant’s telephone number, including area code: (510 ) 745-1700
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
| Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) | |||||
| Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) | |||||
| Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) | |||||
| Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) | |||||
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Trading Symbol(s) | Name of each exchange on which registered | ||||||||||||
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company ☐
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. o
Item 2.02 Results of Operations and Financial Condition.
On February 19, 2026, Ardelyx, Inc. (the “Company”) announced its financial results for the quarter and year ended December 31, 2025. The full text of the press release issued in connection with the announcement is furnished as Exhibit 99.1 to this Current Report on Form 8-K.
The information furnished under this Item 2.02, including Exhibit 99.1 hereto, shall not be considered “filed” under the Securities Exchange Act of 1934, as amended (the “Exchange Act”), nor shall it be incorporated by reference into any future filing under the Securities Act of 1933, as amended (the “Securities Act”), or under the Exchange Act, unless the Company expressly sets forth in such future filing that such information is to be considered “filed” or incorporated by reference therein.
Item 7.01 Regulation FD Disclosure.
On February 19, 2026, the Company will host a conference call to discuss its financial results for the quarter and year ended December 31, 2025. A copy of the earnings presentation that will be used during this conference call is furnished as Exhibit 99.2 to this Current Report on Form 8-K and is incorporated herein by reference.
The information furnished under this Item 7.01, including Exhibit 99.2 hereto, shall not be considered “filed” under the Exchange Act nor shall it be incorporated by reference into any future filing under the Securities Act, or under the Exchange Act, unless the Company expressly sets forth in such future filing that such information is to be considered “filed” or incorporated by reference therein.
Item 9.01 Financial Statements and Exhibits.
(d) Exhibits.
Exhibit No. | Description | |||||||
| 99.1 | ||||||||
| 99.2 | ||||||||
| 104 | Cover Page Interactive Data File (embedded within the Inline XBRL document). | |||||||
SIGNATURE
Pursuant to the requirements of the Securities Exchange Act of 1934, as amended, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| Date: February 19, 2026 | ARDELYX, INC. | |||||||
| By: | /s/ Susan Hohenleitner | |||||||
| Susan Hohenleitner | ||||||||
| Chief Financial Officer | ||||||||

Exhibit 99.1
Ardelyx Reports Fourth Quarter and Full Year 2025 Financial Results and Provides Business Update
IBSRELA revenue grew 73% in 2025 to $274.2 million and total revenues reached $407.3 million
Patient-first XPHOZAH strategy preserved access and drove growth in total dispenses
Development programs for new IBSRELA indication and next-generation NHE3 inhibitor launched
Company is well capitalized to meet current business objectives
Conference call scheduled for 4:30 PM Eastern Time
WALTHAM, Mass., February 19, 2026 - Ardelyx, Inc. (Nasdaq: ARDX), a commercial-stage biopharmaceutical company focused on the development and commercialization of innovative medicines that meet significant unmet medical needs, today reported financial results for the fourth quarter and full year ended December 31, 2025, and provided a business update.
“The results we delivered in 2025 reflect our team’s hard work and disciplined execution to bring our medicines to more patients in need, underscored by significant IBSRELA growth, increased adoption of XPHOZAH and rapid advancement of our clinical development programs,” said Mike Raab, president and chief executive officer of Ardelyx. “As we look to build on this strong momentum, we see 2026 as a pivotal opportunity to further evolve our business into a meaningful enterprise built on a broad, thoughtful portfolio of best-in-class medicines. Our long-term strategy remains clear and unwavering: to deliver novel therapies to patients with significant unmet medical needs and generate sustained value for shareholders.”
IBSRELA® (tenapanor) finishes 2025 with $274.2 million in revenue
Revenue for IBSRELA in 2025 was $274.2 million reflecting 73% growth compared to the $158.3 million reported for the full year 2024. The company recorded $86.6 million in IBSRELA revenue in the fourth quarter of 2025, 61% growth compared to the same period of 2024 and an 11% increase compared to the third quarter of 2025.
Ardelyx expects continued growth in 2026 and beyond, driven by increased depth and breadth of prescribing among target healthcare providers, increased engagement with patients with irritable bowel syndrome with constipation (IBS-C) as well as further improved prescription pull-through.
Ardelyx expects full-year 2026 revenue for IBSRELA to be between $410 and $430 million, representing at least 50% growth compared to 2025. As a result of the significant momentum that IBSRELA has generated, the company expects IBSRELA to achieve $1 billion in annual revenue in 2029, with further growth thereafter.
XPHOZAH® (tenapanor) finishes 2025 with $103.6 million in revenue
Revenue for XPHOZAH in 2025 was $103.6 million reflecting year-over-year growth in total XPHOZAH dispenses, including an increased number of non-Medicare patients on therapy. The company also recorded $27.8 million in revenue in the fourth quarter of 2025.
Ardelyx expects growth in 2026 to be driven by increased clinical conviction and writing among target healthcare prescribers. Ardelyx expects full-year 2026 XPHOZAH revenue to be between $110 and $120 million.
Building a pipeline of important medicines
The company advanced efforts to expand the eligible patient population for IBSRELA to include patients with chronic idiopathic constipation (CIC) and has launched a Phase 3 trial, ACCEL, to assess tenapanor in patients with CIC, dosing the first patient in Q1 2026. Pending the outcome of the Phase 3 trial, Ardelyx intends to submit a Supplemental New Drug Application to the U.S. Food and Drug Administration (FDA) for the CIC indication. Enrollment in ACCEL is expected to be completed by the end of 2026 and topline readout is expected in the second half of 2027.
Ardelyx also launched a development program for RDX10531, a next-generation sodium/hydrogen exchanger 3 (NHE3) inhibitor, in 2025. The company is currently completing pre-clinical development activities in advance of an Investigational New Drug submission to the FDA in the second half of 2026 and initiation of a Phase 1 clinical trial thereafter.

Other Corporate Developments
•United States Patent and Trademark Office issued U.S. Patent No. 12,539,299 titled “Oral Formulations of Tenapanor.” The patent covers the commercial formulations of IBSRELA and XPHOZAH and has an expiration date of November 26, 2042. The patent is listed in the FDA’s Approved Drug Products with Therapeutic Equivalence Evaluations (commonly known as the Orange Book) for both products.
•Four posters were presented at the American Society of Nephrology’s annual Kidney Week, including data from real-world evidence studies demonstrating patient satisfaction and reduction in serum phosphate with XPHOZAH.
•Three posters were presented at the American College of Gastroenterology’s 2025 Annual Meeting supporting the benefits of IBSRELA.
Full Year 2025 Financial Results
• Cash Position: As of December 31, 2025, the company had total cash, cash equivalents and short-term investments of $264.7 million, compared to total cash, cash equivalents and short-term investments of $250.1 million as of December 31, 2024.
• Revenues: Total revenue for the year ended December 31, 2025 was $407.3 million, compared to $333.6 million in total revenue in 2024, driven by increases in IBSRELA revenue.
◦IBSRELA revenue was $274.2 million, compared to $158.3 million in 2024.
◦XPHOZAH revenue was $103.6 million, compared to $160.9 million in 2024.
◦Other revenues, including product supply, licensing and non-cash royalty revenue related to the sale of future royalties, totaled $29.5 million, compared to $14.4 million in 2024.
• R&D Expenses: Research and development expenses were $71.5 million for the year ended December 31, 2025, compared to $52.3 million for the year ended December 31, 2024. The increase was related to investments in recently announced pipeline programs and increased medical engagement with scientific communities.
• SG&A Expenses: Selling, general and administrative expenses were $337.2 million for the year ended December 31, 2025, compared to $258.7 million for the year ended December 31, 2024. The increase was primarily related to increased costs associated with the ongoing commercialization of IBSRELA and XPHOZAH.
• Net Loss: Net loss for the year ended December 31, 2025 was $61.6 million, or $(0.26) per share, compared to net loss of $39.1 million, or $(0.17) per share, for the year ended December 31, 2024. The net loss for the full year 2025 included share-based compensation expense of $49.0 million.
Conference Call Details
The company will host a conference call today, February 19, 2026, at 4:30 PM ET to discuss today’s announcement. To participate in the conference call, please dial (877) 346-6112 (domestic) or (848) 280-6350 (international) and ask to be joined into the Ardelyx call. A live audio webcast and related presentation materials can also be accessed by visiting the Investor page of the company’s website, www.ardelyx.com, and will be available on the website for 30 days following the call.
IMPORTANT SAFETY INFORMATION (IBSRELA)
| WARNING: RISK OF SERIOUS DEHYDRATION IN PEDIATRIC PATIENTS | |||||
| IBSRELA is contraindicated in patients less than 6 years of age; in nonclinical studies in young juvenile rats administration of tenapanor caused deaths presumed to be due to dehydration. Avoid use of IBSRELA in patients 6 years to less than 12 years of age. The safety and effectiveness of IBSRELA have not been established in patients less than 18 years of age. | |||||
CONTRAINDICATIONS
•IBSRELA is contraindicated in patients less than 6 years of age due to the risk of serious dehydration.
•IBSRELA is contraindicated in patients with known or suspected mechanical gastrointestinal obstruction.
WARNINGS AND PRECAUTIONS
Risk of Serious Dehydration in Pediatric Patients
•IBSRELA is contraindicated in patients below 6 years of age. The safety and effectiveness of IBSRELA in patients less than 18 years of age have not been established. In young juvenile rats (less than 1 week old; approximate human age equivalent of less than 2 years of age), decreased body weight and deaths occurred, presumed to be due to dehydration, following oral administration of tenapanor. There are no data available in older juvenile rats (human age equivalent 2 years to less than 12 years).

•Avoid the use of IBSRELA in patients 6 years to less than 12 years of age. Although there are no data in older juvenile rats, given the deaths in younger rats and the lack of clinical safety and efficacy data in pediatric patients, avoid the use of IBSRELA in patients 6 years to less than 12 years of age.
Diarrhea
Diarrhea was the most common adverse reaction in two randomized, double-blind, placebo-controlled trials of IBS-C. Severe diarrhea was reported in 2.5% of IBSRELA-treated patients. If severe diarrhea occurs, suspend dosing and rehydrate patient.
MOST COMMON ADVERSE REACTIONS
The most common adverse reactions in IBSRELA-treated patients (incidence ≥2% and greater than placebo) were: diarrhea (16% vs 4% placebo), abdominal distension (3% vs <1%), flatulence (3% vs 1%) and dizziness (2% vs <1%).
INDICATION
IBSRELA (tenapanor) is indicated for the treatment of Irritable Bowel Syndrome with Constipation (IBS-C) in adults.
Please see full Prescribing Information, including Boxed Warning, for additional risk information.
IMPORTANT SAFETY INFORMATION (XPHOZAH)
CONTRAINDICATIONS
XPHOZAH is contraindicated in:
•Pediatric patients under 6 years of age
•Patients with known or suspected mechanical gastrointestinal obstruction
WARNINGS AND PRECAUTIONS
Diarrhea
Patients may experience severe diarrhea. Treatment with XPHOZAH should be discontinued in patients who develop severe diarrhea.
MOST COMMON ADVERSE REACTIONS
Diarrhea, which occurred in 43-53% of patients, was the only adverse reaction reported in at least 5% of XPHOZAH-treated patients with CKD on dialysis across trials. The majority of diarrhea events in the XPHOZAH-treated patients were reported to be mild-to-moderate in severity and resolved over time, or with dose reduction. Diarrhea was typically reported soon after initiation but could occur at any time during treatment with XPHOZAH. Severe diarrhea was reported in 5% of XPHOZAH-treated patients in these trials.
INDICATION
XPHOZAH (tenapanor), 30 mg BID, is indicated to reduce serum phosphorus in adults with chronic kidney disease (CKD) on dialysis as add-on therapy in patients who have an inadequate response to phosphate binders or who are intolerant of any dose of phosphate binder therapy.
For additional safety information, please see full Prescribing Information.
About Ardelyx
Ardelyx is a commercial-stage biopharmaceutical company focused on the development and commercialization of innovative medicines that meet significant unmet medical needs. Ardelyx has two commercial products approved in the United States, IBSRELA® (tenapanor) and XPHOZAH® (tenapanor). The company’s pipeline includes the Phase 3 development of tenapanor for chronic idiopathic constipation, and RDX10531, a next-generation NHE3 inhibitor with potential application across multiple therapeutic areas. Ardelyx has agreements for the development and commercialization of tenapanor outside of the U.S. Kyowa Kirin commercializes PHOZEVEL® (tenapanor) for hyperphosphatemia in Japan. A New Drug Application for tenapanor for hyperphosphatemia has been approved in China with Fosun Pharma. Knight Therapeutics commercializes IBSRELA in Canada. For more information, please visit https://ardelyx.com/ and connect with us on X (formerly known as Twitter), LinkedIn and Facebook.
Forward Looking Statements
To the extent that statements contained in this press release are not descriptions of historical facts regarding Ardelyx, they are forward-looking statements reflecting the current beliefs and expectations of management made pursuant to the safe harbor of the Private Securities Reform Act of 1995, including Ardelyx’s current expectations regarding: the year in which IBSRELA

will achieve annual U.S. net product sales revenue of $1 billion; the company’s planned label expansion for IBSRELA (tenapanor) to include patients with CIC, pending FDA approval; net product sales revenue for IBSRELA and XPHOZAH for 2026; the company’s ability to deliver sustainable revenue growth, expand its portfolio and deliver meaningful value for shareholders; the timing of the completion of enrollment in the CIC Phase 3 clinical trial and release of topline results; and the timing of an investigational new drug application and initiation of a Phase 1 clinical trial for RDX10531. Such forward-looking statements involve known and unknown risks, uncertainties and other factors that are in some cases beyond our control, that could cause actual outcomes or results to differ materially from those expressed or implied by the forward-looking statements. Such risks and uncertainties include, among others, uncertainties associated with the development of, regulatory process for, and commercialization of drugs in the U.S. and internationally. Ardelyx undertakes no obligation to update or revise any forward-looking statements. For a further description of the risks and uncertainties that could cause actual results to differ from those expressed in these forward-looking statements, as well as risks relating to Ardelyx’s business in general, please refer to Ardelyx’s Annual Report on Form 10-K filed with the Securities and Exchange Commission on February 19, 2026 and its future current and periodic reports to be filed with the Securities and Exchange Commission.
Investor and Media Contact:
Caitlin Lowie
clowie@ardelyx.com

Ardelyx, Inc.
Condensed Balance Sheets
(in thousands)
| December 31, 2025 | December 31, 2024 | ||||||||||
| (Unaudited) | (1) | ||||||||||
| Assets | |||||||||||
| Cash and cash equivalents | $ | 67,999 | $ | 64,932 | |||||||
| Short-term investments | 196,690 | 185,168 | |||||||||
| Accounts receivable | 71,848 | 57,705 | |||||||||
| Prepaid commercial manufacturing | 14,479 | 16,378 | |||||||||
| Inventory | 123,107 | 91,184 | |||||||||
| Property and equipment, net | 2,184 | 1,495 | |||||||||
| Right-of-use assets | 4,795 | 2,380 | |||||||||
| Prepaid and other assets | 20,502 | 16,512 | |||||||||
| Total assets | $ | 501,604 | $ | 435,754 | |||||||
| Liabilities and stockholders’ equity | |||||||||||
| Accounts payable | $ | 19,235 | $ | 16,000 | |||||||
| Accrued compensation and benefits | 19,108 | 14,940 | |||||||||
| Current portion of operating lease liability | 1,479 | 1,562 | |||||||||
| Deferred revenue | 14,905 | 17,918 | |||||||||
| Accrued expenses and other liabilities | 51,218 | 35,665 | |||||||||
| Long-term debt | 202,834 | 150,853 | |||||||||
| Deferred royalty obligation related to the sale of future royalties | 25,876 | 25,527 | |||||||||
| Total stockholders’ equity | 166,949 | 173,289 | |||||||||
| Total liabilities and stockholders’ equity | $ | 501,604 | $ | 435,754 | |||||||
(1) Derived from the audited financial statements included in the Company’s Annual Report on Form 10-K for the year ended December 31, 2024. | |||||||||||

Ardelyx, Inc.
Condensed Statements of Operations
(Unaudited)
(in thousands, except share and per share amounts)
| Three Months Ended December 31, | Twelve Months Ended December 31, | ||||||||||||||||||||||
| 2025 | 2024 | 2025 | 2024 | ||||||||||||||||||||
Revenues | |||||||||||||||||||||||
| Product sales, net | |||||||||||||||||||||||
| IBSRELA | $ | 86,601 | $ | 53,842 | $ | 274,207 | $ | 158,286 | |||||||||||||||
| XPHOZAH | 27,801 | 57,161 | 103,601 | 160,910 | |||||||||||||||||||
| Total product sales, net | 114,402 | 111,003 | 377,808 | 319,196 | |||||||||||||||||||
| Product supply revenue | 9,440 | 4,188 | 15,879 | 11,649 | |||||||||||||||||||
| Licensing revenue | 23 | 22 | 5,088 | 78 | |||||||||||||||||||
| Non-cash royalty revenue related to the sale of future royalties | 1,350 | 916 | 8,545 | 2,692 | |||||||||||||||||||
| Total revenues | 125,215 | 116,129 | 407,320 | 333,615 | |||||||||||||||||||
| Costs and operating expenses | |||||||||||||||||||||||
Cost of sales(1) | 10,849 | 18,264 | 39,537 | 50,556 | |||||||||||||||||||
| Research and development | 22,856 | 13,666 | 71,527 | 52,317 | |||||||||||||||||||
| Selling, general and administrative | 86,411 | 76,074 | 337,233 | 258,692 | |||||||||||||||||||
| Total costs and operating expenses | 120,116 | 108,004 | 448,297 | 361,565 | |||||||||||||||||||
| Income (loss) from operations | 5,099 | 8,125 | (40,977) | (27,950) | |||||||||||||||||||
| Interest expense | (5,759) | (3,967) | (20,102) | (13,006) | |||||||||||||||||||
| Non-cash interest expense related to the sale of future royalties | (1,818) | (1,886) | (8,296) | (7,088) | |||||||||||||||||||
| Other income, net | 2,078 | 2,408 | 8,745 | 9,174 | |||||||||||||||||||
| (Loss) income before provision for income taxes | (400) | 4,680 | (60,630) | (38,870) | |||||||||||||||||||
| Provision for income taxes | 7 | 35 | 969 | 266 | |||||||||||||||||||
| Net (loss) income | $ | (407) | $ | 4,645 | $ | (61,599) | $ | (39,136) | |||||||||||||||
| Net (loss) income per share of common stock - basic and diluted | $ | (0.00) | $ | 0.02 | $ | (0.26) | $ | (0.17) | |||||||||||||||
| Shares used in computing net (loss) income per share - basic | 243,614,026 | 237,370,654 | 241,033,750 | 235,232,927 | |||||||||||||||||||
| Shares used in computing net (loss) income per share - diluted | 243,614,026 | 244,050,606 | 241,033,750 | 235,232,927 | |||||||||||||||||||
(1) Prior year amounts have been reclassified to conform to the current year presentation. | |||||||||||||||||||||||

Q4 and Full Year 2025 Earnings Call February 19, 2026 This presentation is intended for investor purposes only and is not intended for promotional purposes. Exhibit 99.2

2Q4 and Full Year 2025 EarningsThis presentation is intended for investor purposes only and is not intended for promotional purposes. Introduction Caitlin Lowie VP, Investor Relations

3Q4 and Full Year 2025 EarningsThis presentation is intended for investor purposes only and is not intended for promotional purposes. Forward-Looking Statements To the extent that statements contained in this presentation are not descriptions of historical facts regarding Ardelyx, they are forward-looking statements reflecting the current beliefs and expectations of management made pursuant to the safe harbor of the Private Securities Reform Act of 1995, including Ardelyx’s current expectations regarding: the company’s 2026 strategic priorities; the company’s planned label expansion for IBSRELA (tenapanor) to include patients with CIC, pending FDA approval; revenue for IBSRELA and XPHOZAH for 2026; the year in which IBSRELA will achieve annual U.S. net product sales revenue of $1 billion; the timing of the completion of enrollment in the CIC Phase 3 clinical trial, release of topline results, submission of a supplemental new drug application and the timing for when a CIC indication, if approved, may contribute to IBSRELA growth; an d the timing of an investigational new drug application and initiation of a Phase 1 clinical trial for RDX10531. Such forward -looking statements involve substantial risks and uncertainties that could cause Ardelyx's future results, performance or achievements to differ significantly from those expressed or implied by the forward-looking statements. Such risks and uncertainties include, among others, uncertainties associated with the commercialization of drugs and uncertainties regarding the FDA and foreign regulatory processes. Ardelyx undertakes no obligation to update or revise any forward-looking statements. For a further description of the risks and uncertainties that could cause actual results to differ from those expressed in these forward-looking statements, as well as risks relating to Ardelyx's business in general, please refer to Ardelyx’s annual report on Form 10 -K filed with the Securities and Exchange Commission on February 19, 2026, and its future current and periodic reports to be filed with the Securities and Exchange Commission.

Participants Mike Raab President & Chief Executive Officer Eric Foster Chief Commercial Officer Laura Williams, MD, MPH Chief Patient Officer & Interim Chief Medical Officer Sue Hohenleitner, CPA, CMA Chief Financial Officer Caitlin Lowie VP, Investor Relations 4This presentation is intended for investor purposes only and is not intended for promotional purposes. Q4 and Full Year 2025 Earnings

5Q4 and Full Year 2025 EarningsThis presentation is intended for investor purposes only and is not intended for promotional purposes. Opening Remarks Mike Raab President and CEO

6Q4 and Full Year 2025 EarningsThis presentation is intended for investor purposes only and is not intended for promotional purposes. Accelerated IBSRELA growth momentum Executed on XPHOZAH strategy Built a pipeline focused on addressing areas of unmet patient need Delivered strong financial performance Successfully Delivered on Our 2025 Priorities * when excluding Medicare $274.2M 2025 FY Revenue 73% YoY Growth $103.6M 2025 FY Revenue $264.7M Cash & Investments as of 12/31/2025 Positive Cash Flow Generation in 2H 2025 2 Pipeline Programs Launched in 2025 9% YoY Total Dispense Growth

This presentation is intended for investor purposes only and is not intended for promotional purposes. Significantly grow IBSRELA demand Maintain XPHOZAH momentum Further advance a pipeline of innovative medicines Continue delivering strong financial performance Well-Positioned to Execute on Our 2026 Strategic Priorities 2026 Strategic Priorities 7Q4 and Full Year 2025 Earnings Supported by the right leadership, team, strategy and urgency

8Q4 and Full Year 2025 EarningsThis presentation is intended for investor purposes only and is not intended for promotional purposes. Commercial Update Eric Foster Chief Commercial Officer

9Q4 and Full Year 2025 EarningsThis presentation is intended for investor purposes only and is not intended for promotional purposes.This presentation is intended f investor purposes o ly and is not intended for prom tional purposes.
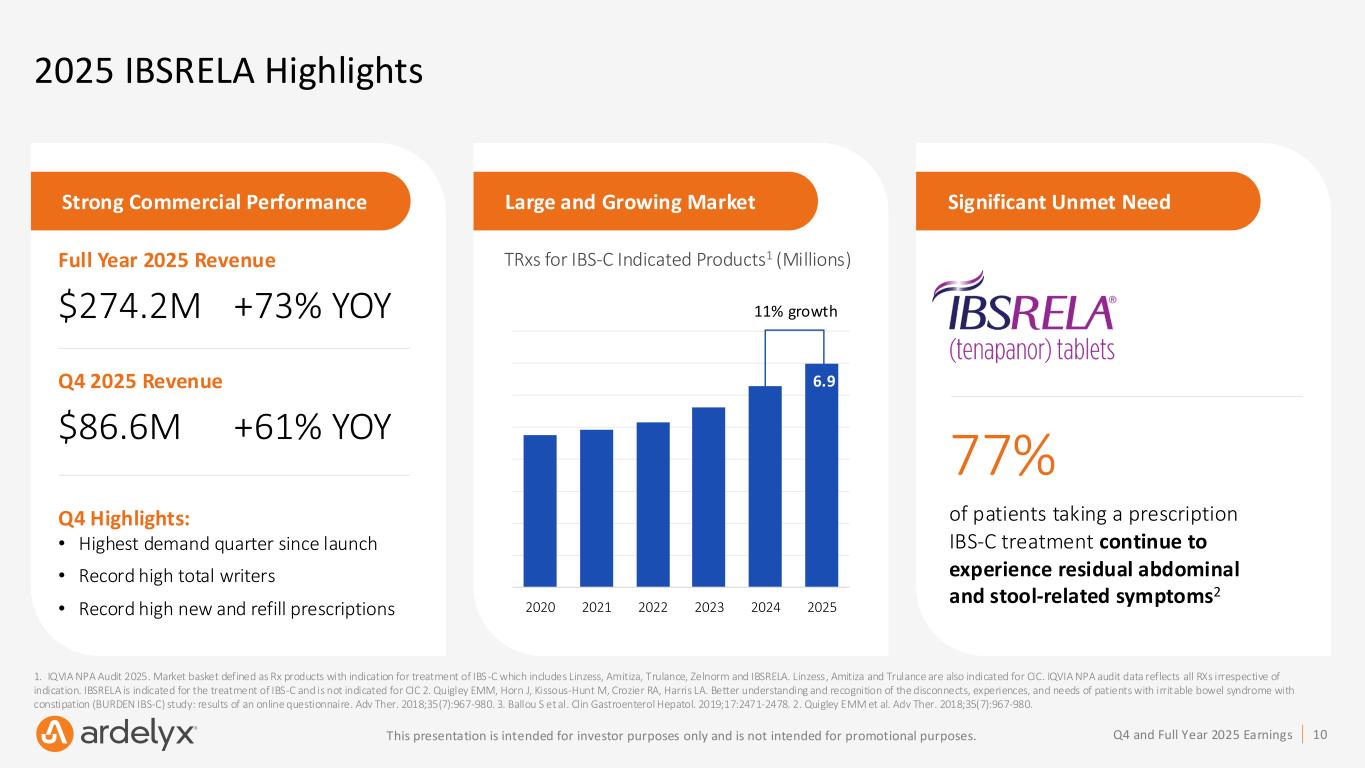
10Q4 and Full Year 2025 EarningsThis presentation is intended for investor purposes only and is not intended for promotional purposes. 2025 IBSRELA Highlights 1. IQVIA NPA Audit 2025. Market basket defined as Rx products with indication for treatment of IBS-C which includes Linzess, Amitiza, Trulance, Zelnorm and IBSRELA. Linzess, Amitiza and Trulance are also indicated for CIC. IQVIA NPA audit data reflects all RXs irrespective of indication. IBSRELA is indicated for the treatment of IBS-C and is not indicated for CIC 2. Quigley EMM, Horn J, Kissous-Hunt M, Crozier RA, Harris LA. Better understanding and recognition of the disconnects, experiences, and needs of patients with irritable bowel syndrome with constipation (BURDEN IBS-C) study: results of an online questionnaire. Adv Ther. 2018;35(7):967-980. 3. Ballou S et al. Clin Gastroenterol Hepatol. 2019;17:2471-2478. 2. Quigley EMM et al. Adv Ther. 2018;35(7):967-980. Strong Commercial Performance Large and Growing Market Significant Unmet Need 77% of patients taking a prescription IBS-C treatment continue to experience residual abdominal and stool-related symptoms2 TRxs for IBS-C Indicated Products1 (Millions) 2020 2021 2022 2023 2024 2025 6.9 11% growth Q4 Highlights: • Highest demand quarter since launch • Record high total writers • Record high new and refill prescriptions Full Year 2025 Revenue $274.2M +73% YOY Q4 2025 Revenue $86.6M +61% YOY
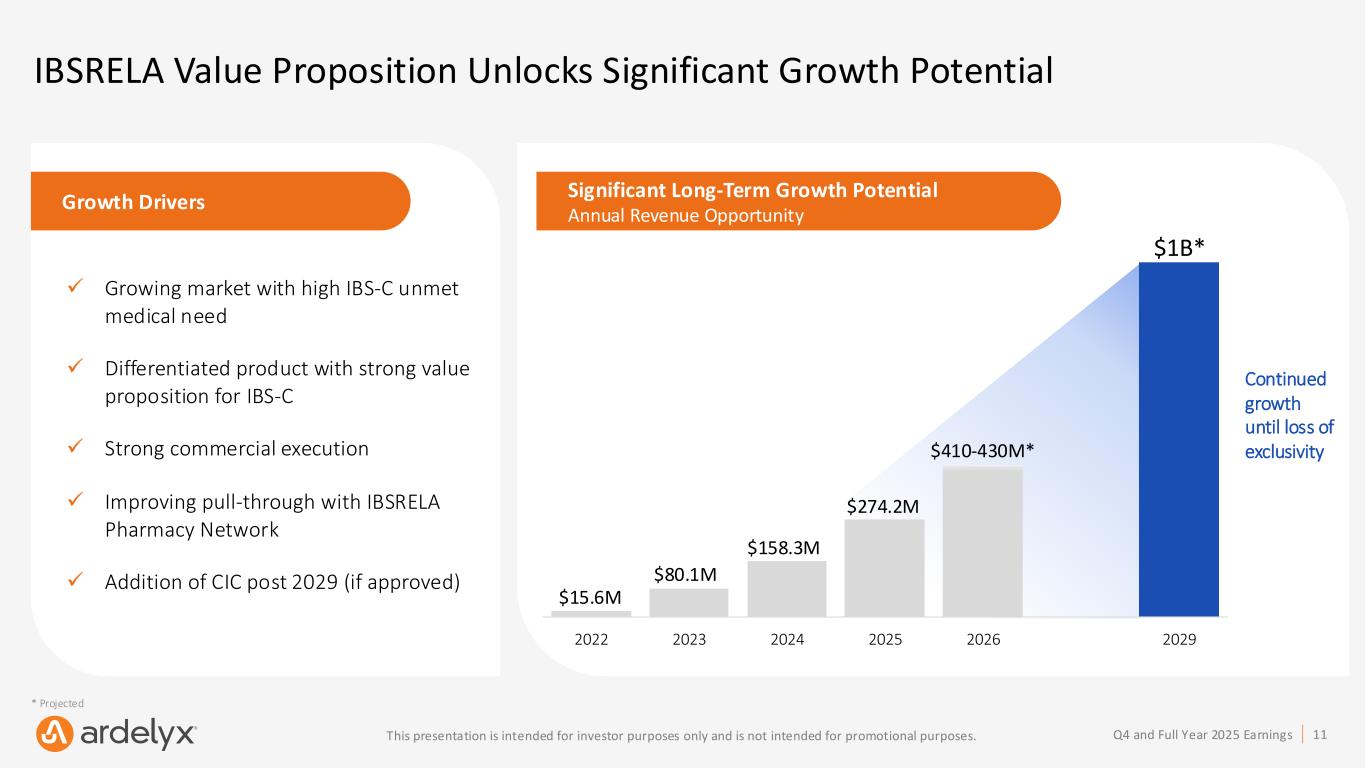
11Q4 and Full Year 2025 EarningsThis presentation is intended for investor purposes only and is not intended for promotional purposes. IBSRELA Value Proposition Unlocks Significant Growth Potential Growth Drivers ✓ Growing market with high IBS-C unmet medical need ✓ Differentiated product with strong value proposition for IBS-C ✓ Strong commercial execution ✓ Improving pull-through with IBSRELA Pharmacy Network ✓ Addition of CIC post 2029 (if approved) Significant Long-Term Growth Potential Annual Revenue Opportunity $158.3M $80.1M $15.6M $274.2M $410-430M* $1B* * Projected 2022 2023 2024 2025 2026 2029 Continued growth until loss of exclusivity

12Q4 and Full Year 2025 EarningsThis presentation is intended for investor purposes only and is not intended for promotional purposes.This presentation is intended for investor purposes only and is not intended for pro otional purposes.
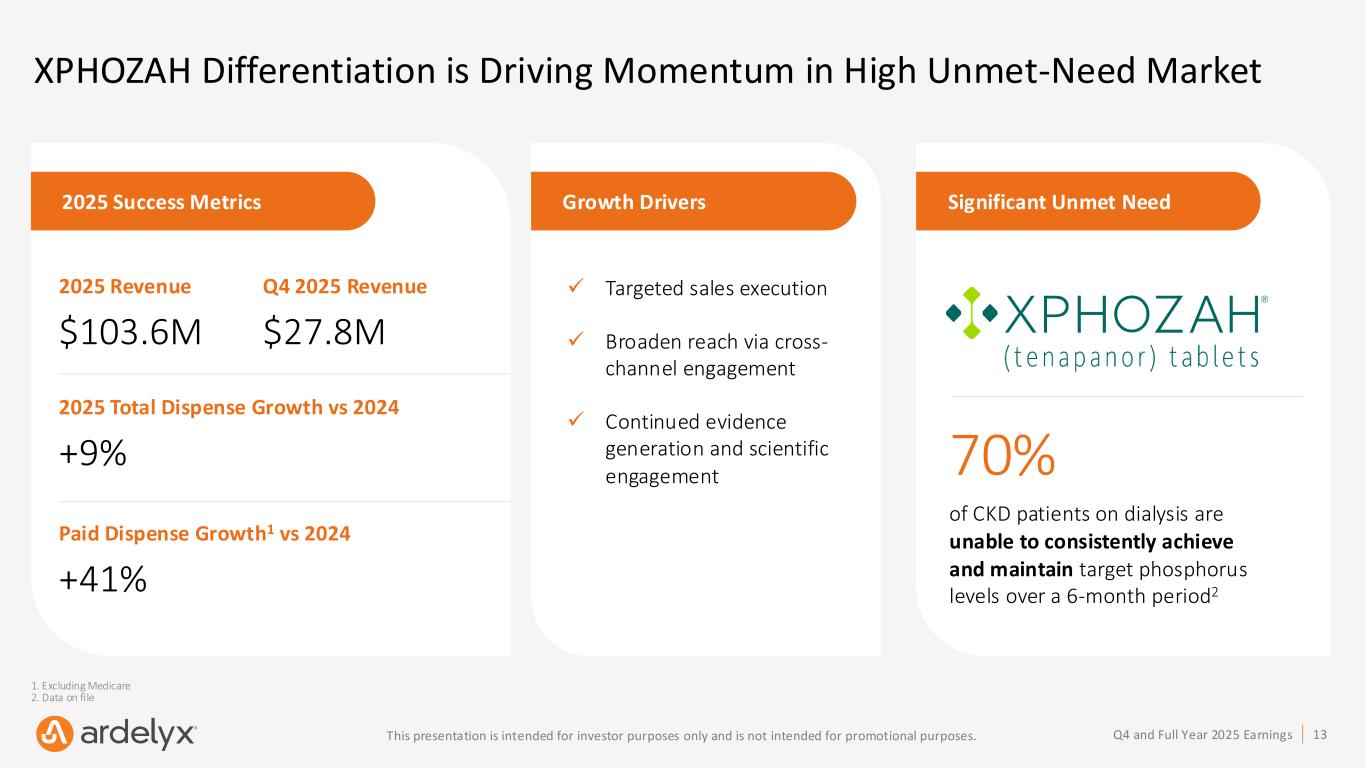
13Q4 and Full Year 2025 EarningsThis presentation is intended for investor purposes only and is not intended for promotional purposes. 2025 Success Metrics 2025 Revenue $103.6M 2025 Total Dispense Growth vs 2024 +9% Paid Dispense Growth1 vs 2024 +41% XPHOZAH Differentiation is Driving Momentum in High Unmet-Need Market Growth Drivers ✓ Targeted sales execution ✓ Broaden reach via cross- channel engagement ✓ Continued evidence generation and scientific engagement Q4 2025 Revenue $27.8M Significant Unmet Need 70% of CKD patients on dialysis are unable to consistently achieve and maintain target phosphorus levels over a 6-month period2 1. Excluding Medicare 2. Data on file

14Q4 and Full Year 2025 EarningsThis presentation is intended for investor purposes only and is not intended for promotional purposes. Development Update Laura Williams, MD, MPH Chief Patient Officer and Interim Chief Medical Officer
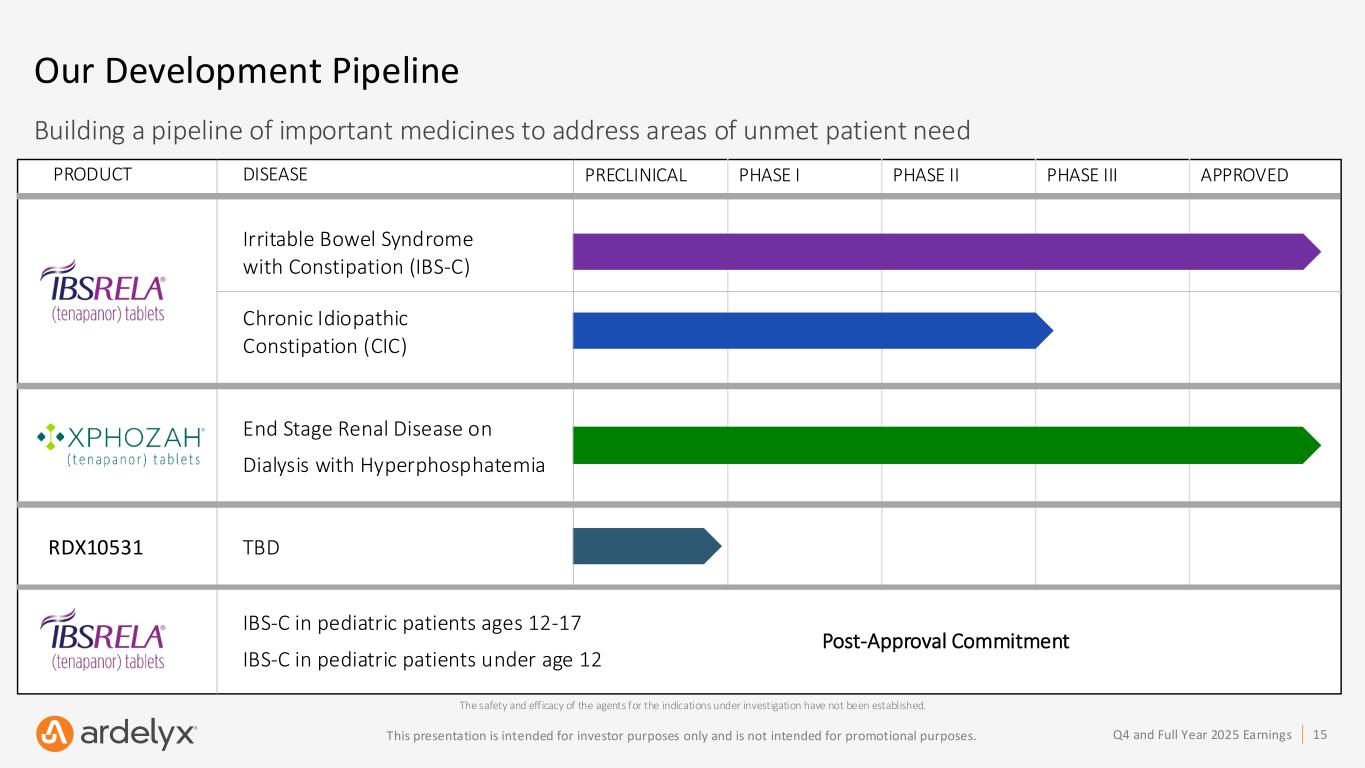
15Q4 and Full Year 2025 EarningsThis presentation is intended for investor purposes only and is not intended for promotional purposes. PRECLINICAL PHASE I PHASE II PHASE III APPROVED Our Development Pipeline The safety and efficacy of the agents for the indications under investigation have not been established. Building a pipeline of important medicines to address areas of unmet patient need PRODUCT DISEASE RDX10531 TBD End Stage Renal Disease on Dialysis with Hyperphosphatemia Irritable Bowel Syndrome with Constipation (IBS-C) Chronic Idiopathic Constipation (CIC) IBS-C in pediatric patients ages 12-17 IBS-C in pediatric patients under age 12 Post-Approval Commitment

16Q4 and Full Year 2025 EarningsThis presentation is intended for investor purposes only and is not intended for promotional purposes. Q4 i Evaluating Tenapanor in Patients with Chronic Idiopathic Constipation (CIC)
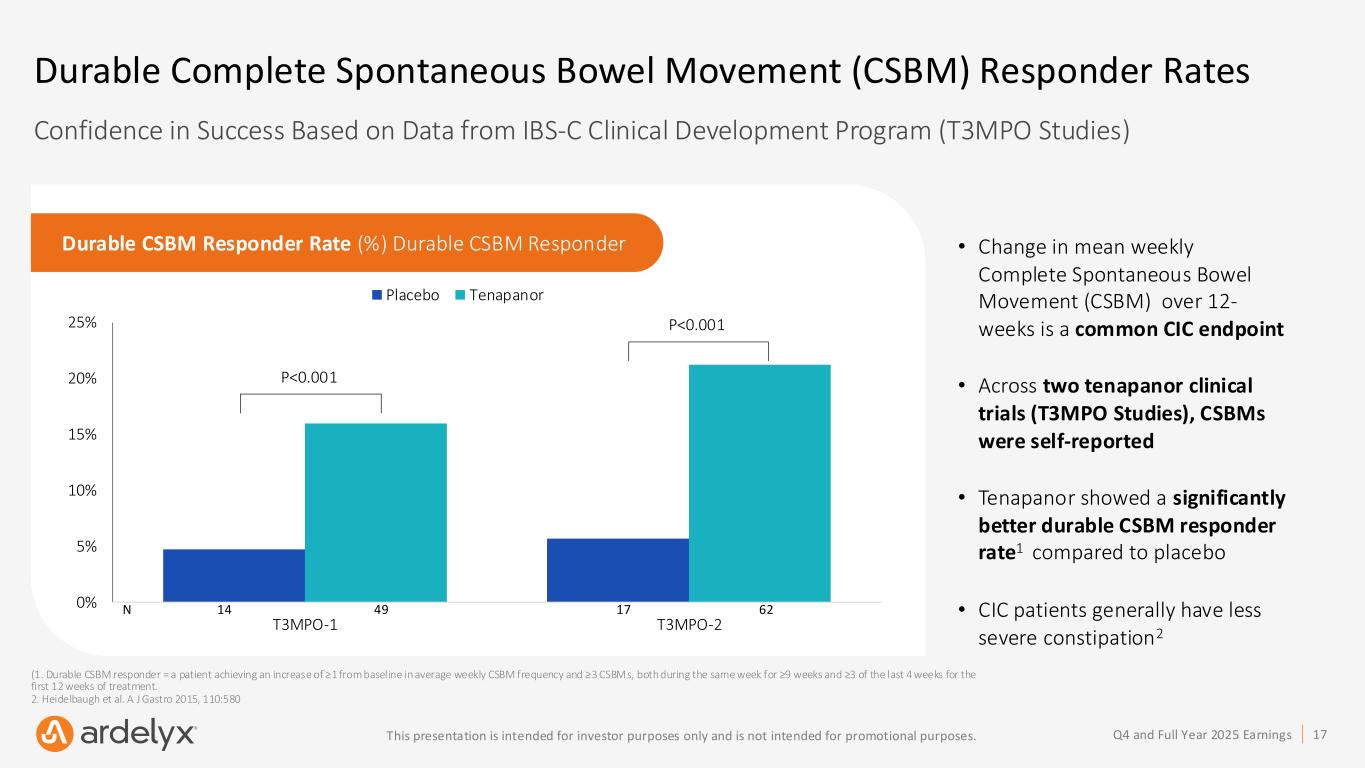
17Q4 and Full Year 2025 EarningsThis presentation is intended for investor purposes only and is not intended for promotional purposes. Durable Complete Spontaneous Bowel Movement (CSBM) Responder Rates Confidence in Success Based on Data from IBS-C Clinical Development Program (T3MPO Studies) • Change in mean weekly Complete Spontaneous Bowel Movement (CSBM) over 12- weeks is a common CIC endpoint • Across two tenapanor clinical trials (T3MPO Studies), CSBMs were self-reported • Tenapanor showed a significantly better durable CSBM responder rate1 compared to placebo • CIC patients generally have less severe constipation2 Durable CSBM Responder Rate (%) Durable CSBM Responder 0% 5% 10% 15% 20% 25% T3MPO-1 T3MPO-2 Placebo Tenapanor P<0.001 P<0.001 (1. Durable CSBM responder = a patient achieving an increase of ≥1 from baseline in average weekly CSBM frequency and ≥3 CSBMs, both during the same week for ≥9 weeks and ≥3 of the last 4 weeks for the first 12 weeks of treatment. 2. Heidelbaugh et al. A J Gastro 2015, 110:580 N 14 49 17 62
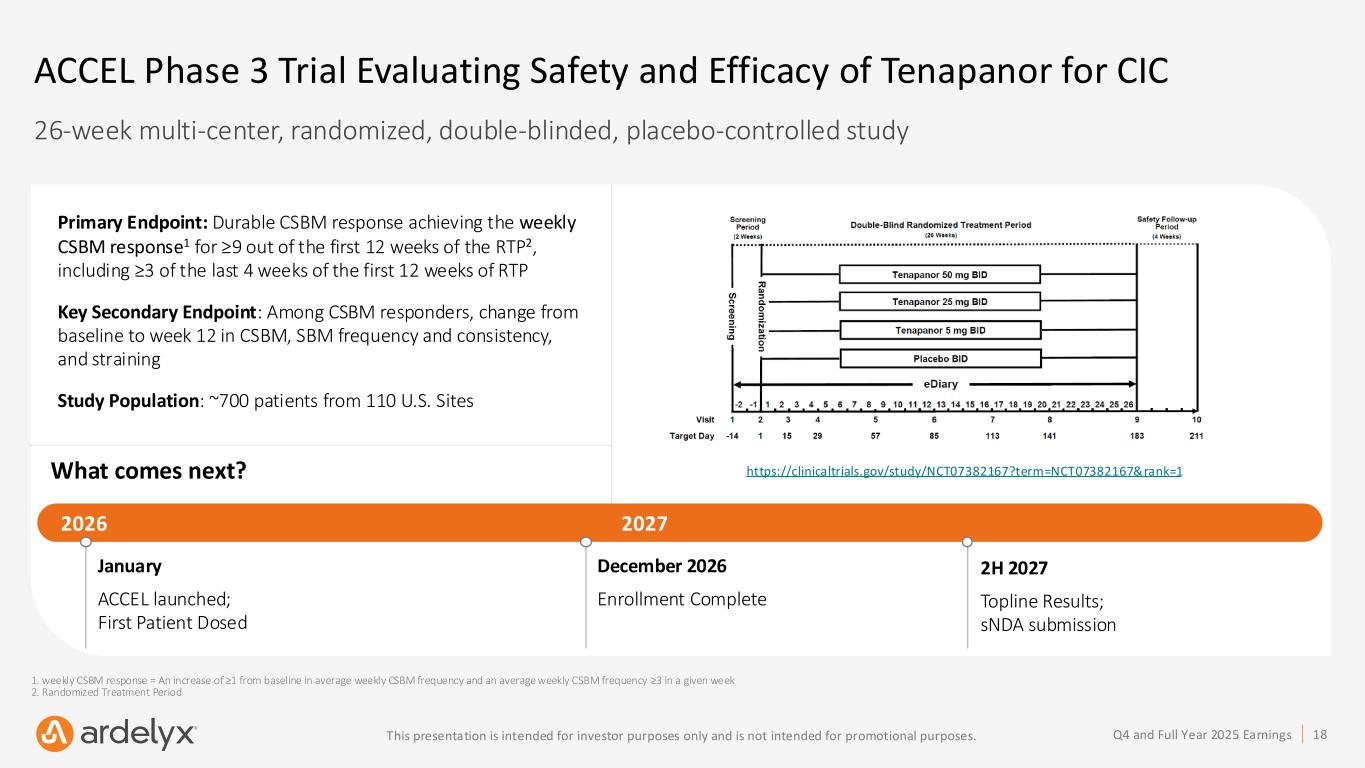
18Q4 and Full Year 2025 EarningsThis presentation is intended for investor purposes only and is not intended for promotional purposes. ACCEL Phase 3 Trial Evaluating Safety and Efficacy of Tenapanor for CIC 26-week multi-center, randomized, double-blinded, placebo-controlled study What comes next? January ACCEL launched; First Patient Dosed December 2026 Enrollment Complete 2H 2027 Topline Results; sNDA submission 2026 2027 Primary Endpoint: Durable CSBM response achieving the weekly CSBM response1 for ≥9 out of the first 12 weeks of the RTP2, including ≥3 of the last 4 weeks of the first 12 weeks of RTP Key Secondary Endpoint: Among CSBM responders, change from baseline to week 12 in CSBM, SBM frequency and consistency, and straining Study Population: ~700 patients from 110 U.S. Sites https://clinicaltrials.gov/study/NCT07382167?term=NCT07382167&rank=1 1. weekly CSBM response = An increase of ≥1 from baseline in average weekly CSBM frequency and an average weekly CSBM frequency ≥3 in a given week 2. Randomized Treatment Period

19Q4 and Full Year 2025 EarningsThis presentation is intended for investor purposes only and is not intended for promotional purposes. Advancing RDX10531, a Next Generation NHE3 Inhibitor Q4 ii t ti i i t f i t l i t i t f ti l .

20Q4 and Full Year 2025 EarningsThis presentation is intended for investor purposes only and is not intended for promotional purposes. • NHE3 is an antiporter expressed on the apical surface of the small and large intestines and kidney, and is responsible for absorbing the majority of ingested sodium, maintaining fluid and pH balance • Preclinical data demonstrates that RDX10531 is a highly potent, highly soluble molecule with the potential for broad application across multiple therapeutic areas • The company is currently conducting Investigational New Drug (IND)-enabling activities and plans to submit an IND in 2H 2026 Ardelyx Has the Only Approved Modulator of This Pathway on the Market Tenapanor
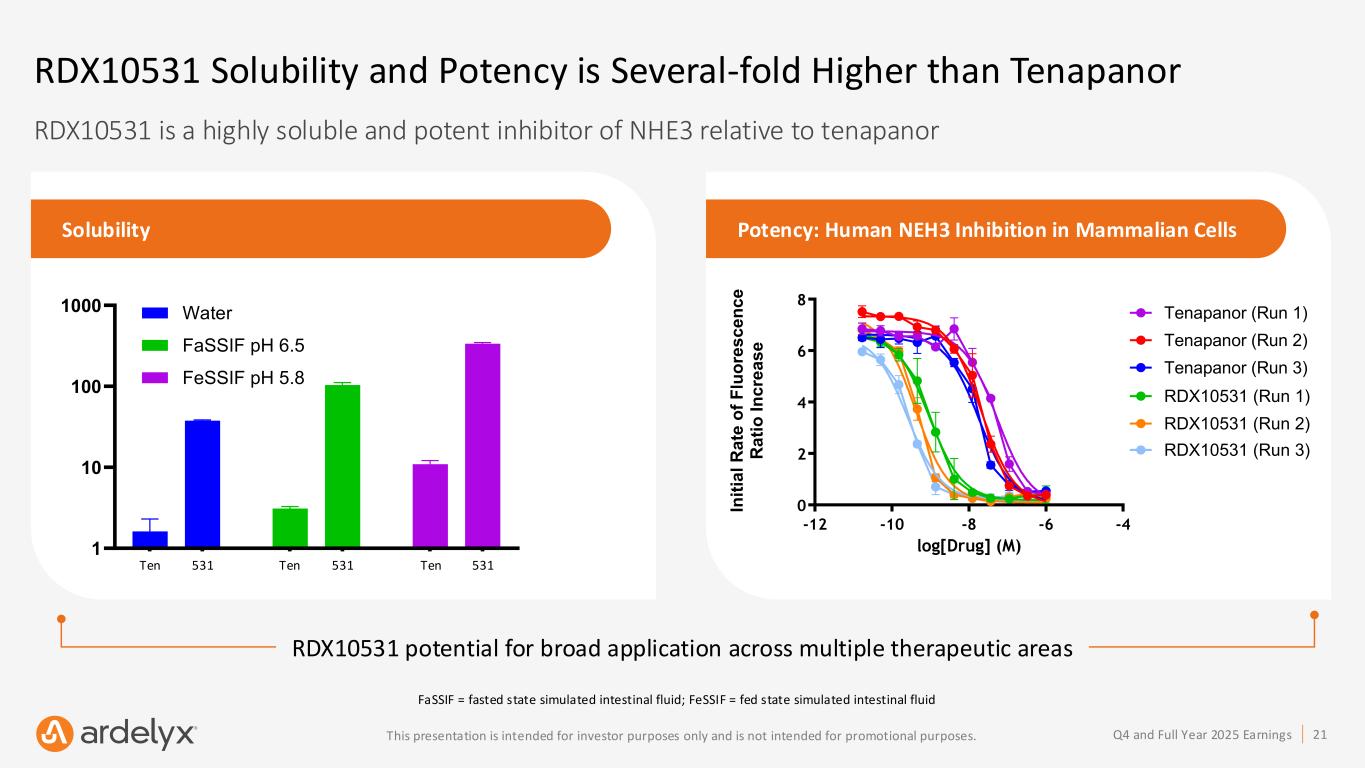
21Q4 and Full Year 2025 EarningsThis presentation is intended for investor purposes only and is not intended for promotional purposes. RDX10531 potential for broad application across multiple therapeutic areas RDX10531 is a highly soluble and potent inhibitor of NHE3 relative to tenapanor RDX10531 Solubility and Potency is Several-fold Higher than Tenapanor Solubility Potency: Human NEH3 Inhibition in Mammalian Cells Ten ap an or R D X 10 53 1 Ten ap an or R D X 10 53 1 Ten ap an or R D X 10 53 1 1 10 100 1000 Solubility C o n c e n tr a ti o n ( µ M ) Water FaSSIF pH 6.5 FeSSIF pH 5.8 Human NHE3 Inhibition in Mammalian Cells -12 -10 -8 -6 -4 0 2 4 6 8 log[Drug] (M) In it ia l R a te o f F lu o re s c e n c e R a ti o I n c re a s e Tenapanor (Run 2) Tenapanor (Run 1) Tenapanor (Run 3) RDX10531 (Run 1) RDX10531 (Run 2) RDX10531 (Run 3) Ten 531 Ten 531 Ten 531 FaSSIF = fasted state simulated intestinal fluid; FeSSIF = fed state simulated intestinal fluid

22Q4 and Full Year 2025 EarningsThis presentation is intended for investor purposes only and is not intended for promotional purposes. Financial Performance Sue Hohenleitner, CPA, CMA Chief Financial Officer
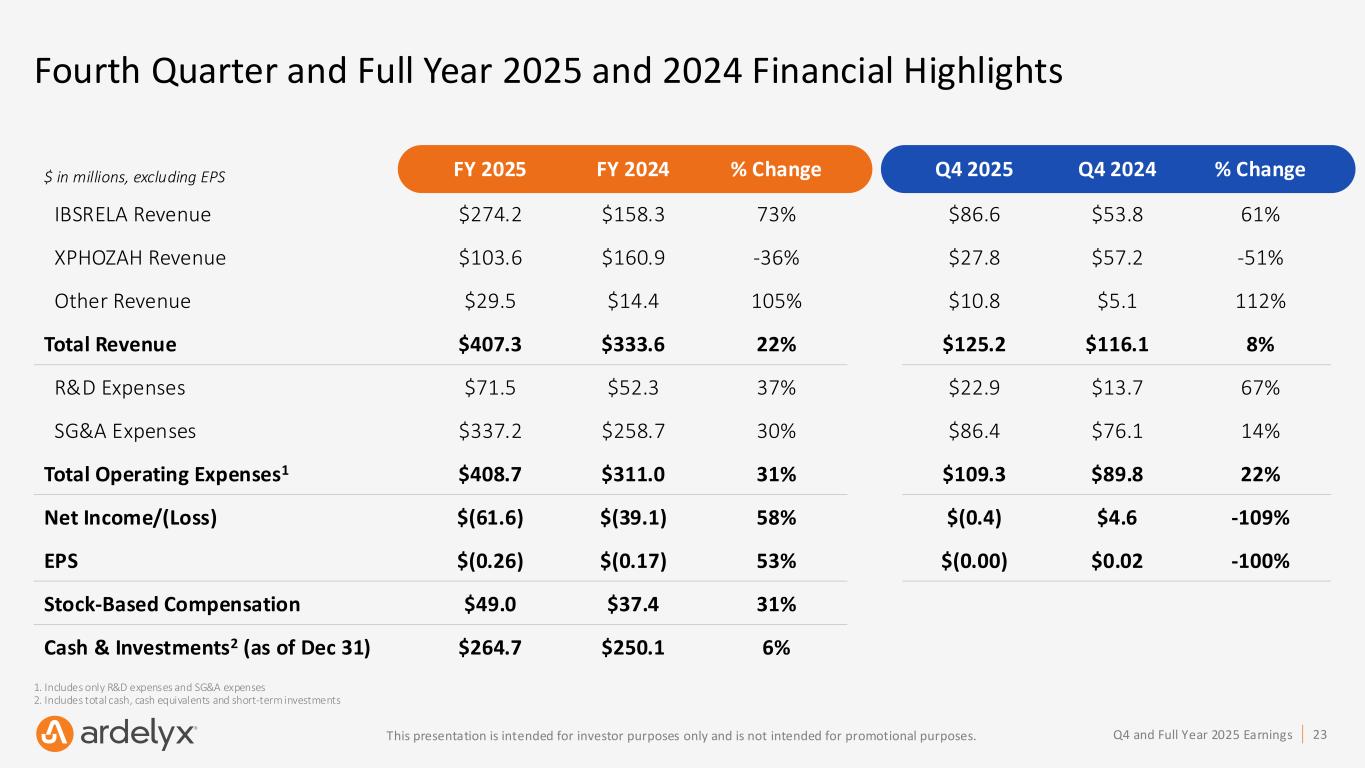
23Q4 and Full Year 2025 EarningsThis presentation is intended for investor purposes only and is not intended for promotional purposes. Fourth Quarter and Full Year 2025 and 2024 Financial Highlights $ in millions, excluding EPS FY 2025 FY 2024 % Change Q4 2025 Q4 2024 % Change IBSRELA Revenue $274.2 $158.3 73% $86.6 $53.8 61% XPHOZAH Revenue $103.6 $160.9 -36% $27.8 $57.2 -51% Other Revenue $29.5 $14.4 105% $10.8 $5.1 112% Total Revenue $407.3 $333.6 22% $125.2 $116.1 8% R&D Expenses $71.5 $52.3 37% $22.9 $13.7 67% SG&A Expenses $337.2 $258.7 30% $86.4 $76.1 14% Total Operating Expenses1 $408.7 $311.0 31% $109.3 $89.8 22% Net Income/(Loss) $(61.6) $(39.1) 58% $(0.4) $4.6 -109% EPS $(0.26) $(0.17) 53% $(0.00) $0.02 -100% Stock-Based Compensation $49.0 $37.4 31% Cash & Investments2 (as of Dec 31) $264.7 $250.1 6% 1. Includes only R&D expenses and SG&A expenses 2. Includes total cash, cash equivalents and short-term investments
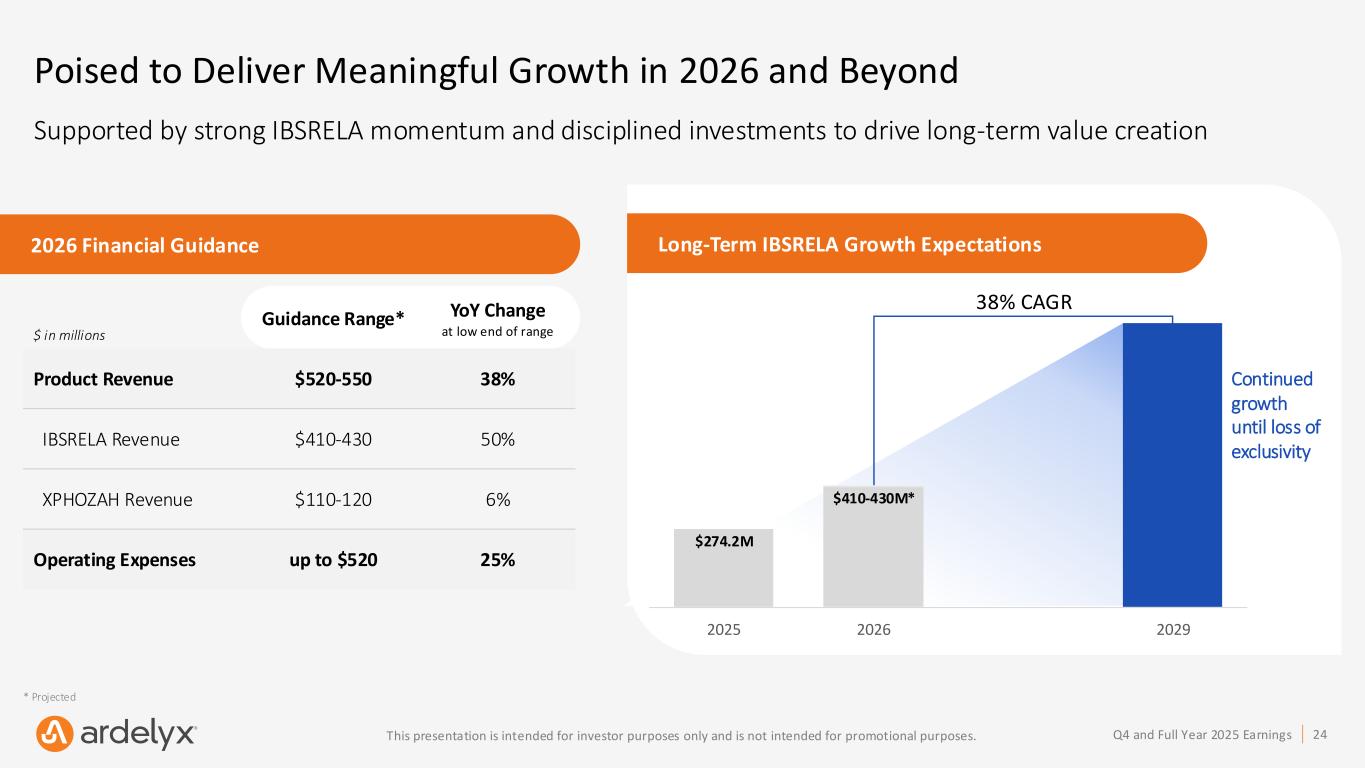
24Q4 and Full Year 2025 EarningsThis presentation is intended for investor purposes only and is not intended for promotional purposes. Poised to Deliver Meaningful Growth in 2026 and Beyond Supported by strong IBSRELA momentum and disciplined investments to drive long-term value creation $ in millions Guidance Range* YoY Change at low end of range Product Revenue $520-550 38% IBSRELA Revenue $410-430 50% XPHOZAH Revenue $110-120 6% Operating Expenses up to $520 25% Long-Term IBSRELA Growth Expectations 38% CAGR 2026 Financial Guidance * Projected $1B* 2025 2026 2029 $274.2M $410-430M* Continued growth until loss of exclusivity

25Q4 and Full Year 2025 EarningsThis presentation is intended for investor purposes only and is not intended for promotional purposes. Closing Remarks Mike Raab President and CEO

26Q4 and Full Year 2025 Earnings Q&A This presentation is intended for investor purposes only and is not intended for promotional purposes.

Thank You This presentation is intended for investor purposes only and is not intended for promotional purposes.
